A new generation of high-density multielectrode arrays (MEAs) now allows the characterisation of neural population activity in unprecedented detail. Here, we present several methods developed for the analysis of such recordings from the neonatal mouse retina with the 4,096 channel Active Pixel Sensor (APS) MEA, which allows near-cellular resolution recordings (electrode diameter of 21um ; 42um pitch; Berdondini et al., Lab on a Chip, 2009,
http://dx.doi.org/10.1039/B907394A). Neural activity in the neonatal mouse retina consists of spontaneous, correlated bursts of neighbouring retinal ganglion cells (RGCs), resulting in propagating waves. Retinal waves undergo several developmental stages, early waves are mediated by gap junctions (Stage I), followed by lateral connections between cholinergic starburst amacrine cells (late gestation to P9; Stage II), and finally by glutamatergic transmission (P10; Stage III) before they disappear at the time of eye opening (P12). Despite such major developmental changes in network connectivity, the low spatial resolution of conventional MEAs has, so far, prevented a systematic observation of concomitant changes in population activity. Here, we report several important developmental changes in wave dynamics as revealed by pan-retinal high-density recordings. A challenge in analysing such data is the quantitative analysis of the spiking activity of thousands of active channels, which typically show considerable heterogeneity. As histogram-based approaches are impractical for such data sets, we have developed an efficient, real-time capable method based on the representation of spike trains as random walks, which allows for reliable classification into bursting, regular or randomly active neurons. After detecting bursts of propagating activity (cf. Hennig et al, 2012,
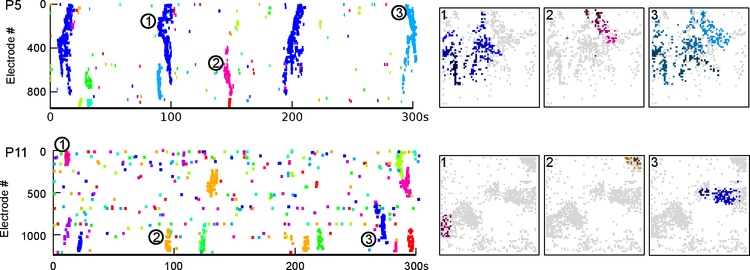
http://dx.doi.org/10.1523/JNEUROSCI.3112-11.2011), waves were grouped and analysed with alpha-shapes, yielding measures of the size and recruitment within wave areas. We found that Stage II waves showed low recruitment and highly variable sizes, becoming larger and denser up to P6, and smaller and slower at P8-9. Stage III waves were denser and more restricted spatially. Finally, wave trajectories were obtained and similarities within recordings analysed by clustering them. This revealed that Stage II waves propagated slowly with a high degree of randomness, while Stage III waves were faster and follow few repetitive, non-random propagation patterns.

 Latest news for Neuroinformatics 2011
Latest news for Neuroinformatics 2011 Follow INCF on Twitter
Follow INCF on Twitter